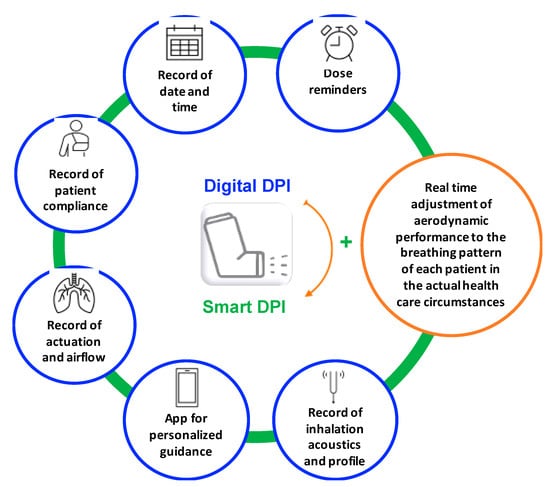
Pharmaceutics | Free Full-Text | Dry Powder Inhalers in the Digitalization Era: Current Status and Future Perspectives

Budesonide/formoterol MDI with co-suspension delivery technology in COPD: the TELOS study | European Respiratory Society